Partnerships

Power up your business with FlexBeam
In the pursuit of accelerated recovery for your patients, you have to decide: which tools should you use? FlexBeam is the most powerful infrared therapy device of its kind. Find out how FlexBeam can help revolutionize your business.

Power up your business with FlexBeam
In the pursuit of accelerated recovery for your patients, you have to decide: which tools should you use? FlexBeam is the most powerful infrared therapy device of its kind. Find out how FlexBeam can help revolutionize your business.

Innovative. Technologically advanced. Affordable.
Recharge Health developed FlexBeam to help therapists and trainers provide faster and more permanent recovery to patients, whether they’re average people or professional athletes.
Innovative. Technologically advanced. Affordable.
Recharge Health developed FlexBeam to help therapists and trainers provide faster and more permanent recovery to patients, whether they’re average people or professional athletes.
Incorporate FlexBeam into your practice and get a device that:
Place directly on skin, wrap around the injury for deep light stimulation, enhancing outcomes for pain and injury.
FlexBeam is a non-invasive treatment, with highly effective results and no known side effects. It provides accelerated recovery from any type of injury (up to 30%, as measured by internal evaluation against sham devices and feedback from the professional users).
Enables early rehabilitation, especially in cases where pain prevents movements.
Apply to area of concern for 10 minutes, even while implementing other therapies. You can apply FlexBeam to 3-4 areas of the body in one session. If working with two devices at once, this can be doubled within 30 minutes.
Use during rehab exercises for increased effectiveness.
Seamlessly integrate into daily activities like cooking, gaming, or watching TV, enhancing patient convenience and compliance.
Enables therapy continuation between appointments, ensuring consistency and progress beyond the clinic.
Recharge Health developed FlexBeam to help therapists and trainers provide faster and more permanent recovery to patients, whether they’re average people or professional athletes.
Partner with us to:
1. Enhance your Patients’ Recovery Solutions
FlexBeam offers innovative infrared technology that accelerates muscle and tissue healing by 20% to 30% – meaning patients can save 1/3 of their visits, and athletes can recover faster from injuries, intense workouts, or competitions, allowing them to maintain peak performance levels.
2. Expand your Revenue Streams
Partnering with Recharge Health and FlexBeam opens up additional revenue streams for sports and physical therapists and performance trainers. Through the affiliate, reseller, FlexBeam rental or distribution programs, you’ll have the ability to earn additional income or commissions.
This not only adds a passive income stream but also provides an opportunity to diversify service offerings and attract new patients interested in the most advanced and relevant recovery technologies.
3. Access Support and Resources
Recharge Health offers comprehensive support and resources, including 24/7 global customer service support, repair centers, marketing and training materials, and the most relevant scientific literature supporting our claims.
Sports/physical therapists and performance trainers can leverage these resources to educate patients about the benefits of near infrared light therapy and FlexBeam, and effectively integrate it into your practice.
If certification in use of Photobiomodulation devices is required: we can offer assistance to provide certified training in Photobiomodulation with a CPD accredited provider, which will in turn help to insure therapy in your practice. For more information contact us.
FlexBeam applications
| For Physical Therapists & Doctors: | For Sport Trainers & Coaches: |
|---|---|
| Support Rehabilitation and Speeds Recovery | Optimize Sports Performance |
| Post-operative | Provide pre-conditioning |
| Post-traumatic | Reduce fatigue |
| Orthopedic | Enhance strength and endurance |
| Neurological | Relieve stress and promoting relaxation |
| Women’s health | Improve sleep |
| Chronic pain management | Regulate circadian rhythm |
| Amputee rehabilitation | Quicker recovery from injuries |
| Occupational health | Expedited recovery from detraining |
| Safe for all ages (children 12 and above) | Safe for animal rehabilitation and recovery |

Head Physiotherapist, Consensio, Norway
Thomas Jensen
“FlexBeam has significantly shortened treatment times and produced much better results overall for our patients. The device is very, very effective. I recommend many clients to buy one for easy home use.”

Major League Baseball Head Athletic Trainer, Milwaukee Brewers
Brad Epstein
“We currently use FlexBeam as both a pre-activity and recovery modality. The ability to wrap it around an extremity such as shoulder and elbow are key for us.”

Physiotherapist, URI Soča, Slovenia
Primož Hostnik
“I use FlexBeam daily, with almost every patient I treat. It gives them energy, enhances their blood flow, and they feel it almost instantly.”

Head Athletic Coach, 1. FSV Mainz 05
Axel Busenkell
“This technology is a real game changer. Many players have already bought their own and also use it to improve strength and performance.”

Vagus Clinic Ltd., Canada
Dr. Stephanie Canestraro
“I work with everyone, from really sick people that have Lyme and MS, to elite players in the NHL. FlexBeam is one of my big recommendations.”
How does the technology behind FlexBeam work?
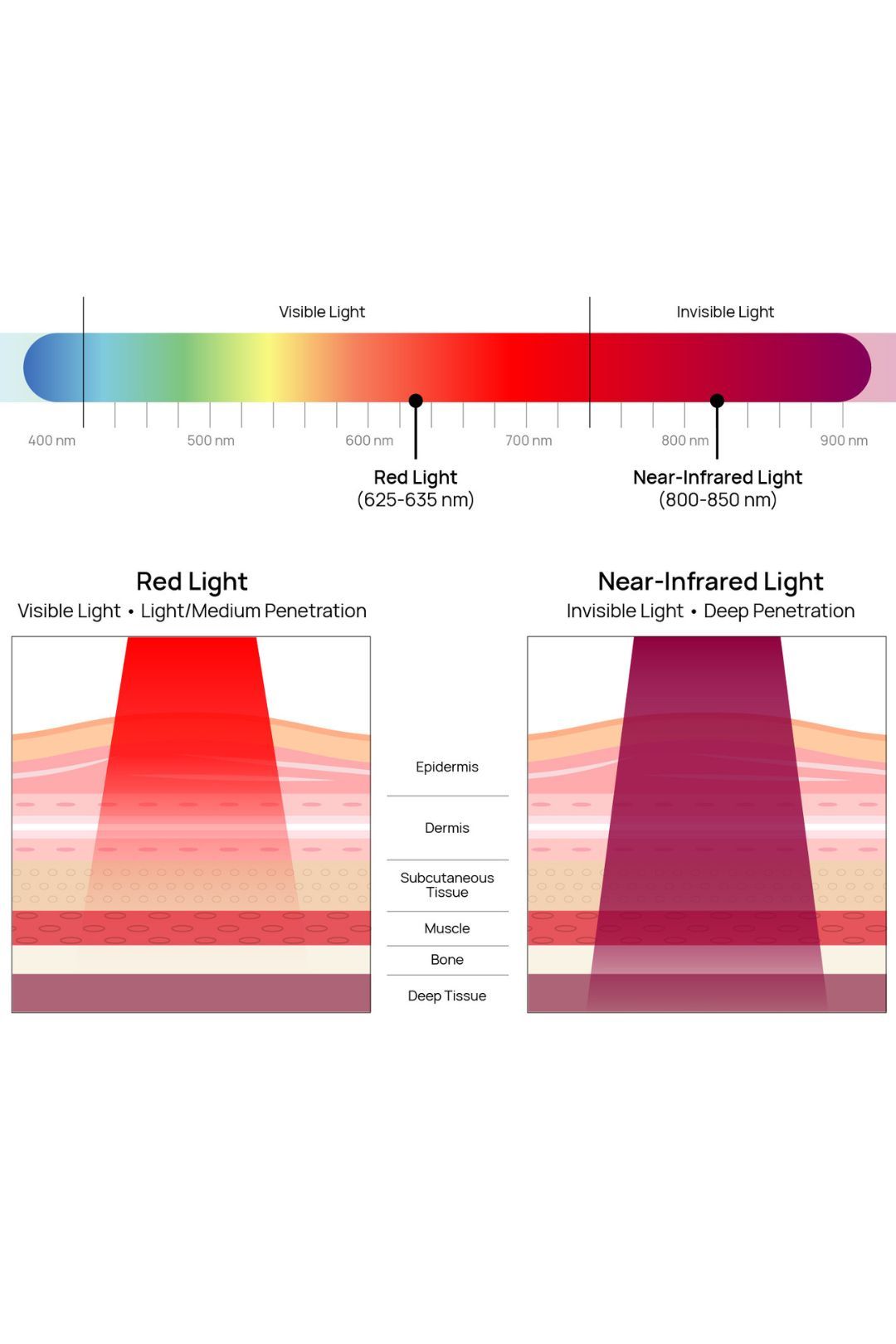
- Red and near-infrared wavelengths, also known as “red light,” are employed in Red Light Therapy (RLT). Unlike thermal mechanisms, RLT relies on a photochemical effect. This effect, known as Photobiostimulation, triggers cellular-level physiological changes called Photobiomodulation (PBM). PBM operates at low energies, in contrast to high-powered medical lasers used for procedures such as cauterizing blood vessels or tissues in hospitals.
- The Cytochrome C Oxidase (CCO), is a chromophore that absorbs light between 570nm to 850nm. FlexBeam, operating at 625-635nm in red and 810-830nm in near-infrared, aligns with the primary absorption range in the IV complex of the respiratory chain, directly stimulating energy processes in irradiated tissues’ cellular structures.
- RLT prompts Nitric Oxide (NO) release from mitochondria. NO inhibits CCO, displacing oxygen in stressed cells. Red and near-infrared light restore CCO function by displacing NO, enhancing cellular respiration, microcirculation and cell-signaling pathways.
- Red light activates various receptors beyond mitochondria, like NHDA, ion channels, opsins, flavins, and flavoproteins. This boosts electron transfer across the mitochondrial membrane, elevating charge levels through a cascade of reactions.
- ROS (Reactive Oxygen Species) released post-RLT initiate signaling pathways, boosting gene expression for synthesis, cell migration, and anti-inflammatory responses. Stem cells are especially receptive. Light restructures water into an Exclusion Zone (EZ), heightening cell charge, affecting the membrane and capacitance.
- RLT stimulates the release of Melatonin (MT), a hormone with potent antioxidant properties. Pineal MT regulates seasonal rhythms, metabolism, immune response, and more. Its high tissue concentrations are crucial for antioxidant and anti-inflammatory effects, vital for mitochondrial function. Melatonin is a potential mediator of RLT therapeutic effects.
How does the body heal faster with FlexBeam?
The human body undergoes three stages of healing: inflammation, cellular proliferation, and remodeling.
FlexBeam can be used during each of these stages to support the body’s natural healing responses.
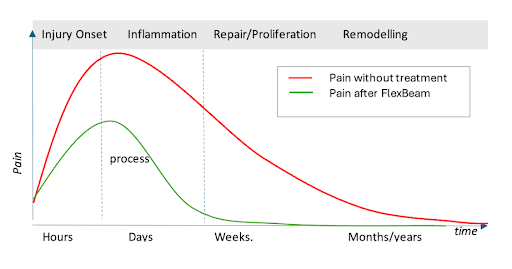
The effect of FlexBeam on pain
Healing with FlexBeam, continued:
With high metabolic rate, this stage is ENERGY demanding.
Red and infrared light therapy enhance local circulation of nitric oxide (NO), electron transport, stimulates mitochondrial respiration, and increases ATP production. Concurrently, there is an elevation in Mitochondrial Membrane Potential (MMP), resulting in a tangible increase in energy generation. Galvanotaxis facilitates active healing at the cellular level, while active angiogenesis contributes to faster healing.
During this stage, there is an integration of damaged tissues and scar formation.
Red and infrared light therapy activates stem precursor cells, satellite cells and enables cellular proliferation, increasing collagen production and decreasing collagen degradation. There is a proliferation of fibroblasts and faster conversion from Collagen III to Collagen I.
During this stage, fibroblasts, myoblasts, osteoblasts, and other stem cells are activated to repair damage, form new functional tissues, and contract and reorganize scars. Red and infrared light therapy are known to accelerate the remodelling of scar tissue and promote the regeneration of nerve cells.

Get started today
Join the growing community of physical therapists, businesses, and other partnerships sharing the power of red and infrared light with their clients, patients, and customers. Learn more about our tiered approach to wholesale discounts. Email us at: [email protected] or click the button below.
Get started today
Join the growing community of physical therapists, businesses, and other partnerships sharing the power of red and infrared light with their clients, patients, and customers. Learn more about our tiered approach to wholesale discounts. Email us at: [email protected] or click the button below.
Order nowAbout FlexBeam



The device, in detail
FlexBeam is a portable Photobiomodulation (PBM) device that emits non-coherent red and near-infrared light via LEDs. FlexBeam is designed specifically to help boost wellness.
FlexBeam dimensions are 35cm x 13.5cm x 4cm. The 3 light modules plus one battery module are connected by a flexible silicon material allowing the device to be wrapped around a limb. In addition, it can be or secured flat against the body by using the provided straps, where necessary. This enables treatment on any area of the body and, in the case of wrapping around a limb, treatment from multiple angles.
The device is comprised of 6 Near infrared (NIR) LEDs at 810-830 nm (3 modules, 2 LEDs/module) and 3 red LEDs at 625-635nm (3 modules, 1 LED/module). The LEDs are high quality with a 90-245V AC input power supply with 15V, 2.4A output. Peak wavelength 630 nm in red and 815 nm in near-infrared. When in contact, it irradiates 9 cm2 of skin per module, a total of 81 cm2.
The key technical parameters of FlexBeam were calculated with a consideration of fluence, dose of energy and bi-phasic dose response. When in contact with skin, FlexBeam has a maximum power output of 100mW/cm2 in the NIR LEDs and 50mW/cm2 in the red LEDs. The device operates in pulsing mode with pulsing frequencies between 10-1000 Hz and duty cycle of 10-100% with 10-minute intervention time. All materials in direct contact with the skin meet ISO 10993 requirements for bio-compatibility for cytotoxicity, sensitisation, and irritation.
FlexBeam is powered by a rechargeable Li-ion battery contained in the battery module with one charge lasting for up to 1.5 hours. This module also contains the user interface for turning on and shutting down the device, program selection and indication of battery life.
The device contains an active fan cooling system to regulate temperature and automatic shutdown at the completion of each treatment. It is also programmed to shut down in the case of overheating or overcurrent faults to ensure user safety.
The FlexBeam is intended to be used directly on the skin, anywhere on the body. It is not designed to treat the head. It’s an over-the-counter device intended for the temporary relief of minor muscle and joint pain, arthritis and muscle spasm; relieving stiffness; temporary relaxation of muscle tissue and to temporarily increase local blood circulation.
FlexBeam emits non-coherent red and near-infrared (NIR) light, and its effects are accomplished through an optimization of mitochondria functioning and an increase in microcirculation stimulated by this light.
Additional associated benefits are faster recovery rate from physical performance, increased energy level, and improvement of general wellbeing and sleep.
It’s not just red light therapy. It’s FlexBeam.
FlexBeam is a complete, wearable red light therapy system designed to make advanced recovery practical, personal, and repeatable. By combining deeper light penetration, precise targeted placement, and consistent, hands-free use, it delivers support where your body needs it. Engineered for real bodies and real routines, to be used in the comfort of home.

Power output
Optical power output: up to 5.5W
Cooling
Active cooling fans built in to use max power LEDs.
Battery
Lithium-ion battery that holds 5-8 sessions (50 - 80 minutes) on a single charge.
Presets
3 easy-to-use presets to vary depth of treatment. 10 minute programs, auto-shutoff.
Bands
Easy-to-use velcro bands for fastening on the body.
Wavelengths
Synergistic wavelengths: 610-650nm (red) and 800-850nm (NIR) No EMFs (electro-magnetic field radation).
Energy density
Energy density / dose: up to 110mW/cm2
Clinically Relevant Wavelengths
Uses specific red and near-infrared wavelengths selected to support mitochondrial function and recovery.
Deeper Tissue Penetration
Engineered to deliver light beyond the surface, supporting muscles, joints, and connective tissue where recovery actually happens.
Targeted, Body-Specific Placement
Flexible design allows precise positioning on knees, shoulders, back, hips, and more, not just general exposure.
Hands-Free, Wearable Design
No standing in front of panels. No holding devices. Use it while you work, relax, or recover.
Consistent Dosing by Design
Built for repeatable sessions and structured protocols, because results depend on consistency.
Built for Real Life
Designed to integrate into everyday routines. At home, at work, or after training.
Regulatory compliance
Recharge Health and FlexBeam® achieved EC and CE Electrical Safety Compliance in accordance with the ISO 13485 medical device quality standard, and is registered with the US FDA, Saudi FDA and Thai FDA authorities. In the USA, FlexBeam® is considered a Class II medical device exempted from FDA approval. This means it is a self-certified device that complies with the highest safety and quality standards, and is open for FDA inspection anytime.
In the rest of the world FlexBeam is distributed and sold as a wellness device.


🔌 IEC 60601-1:2012 (Ed. 3.1). Medical Electrical Equipment – General Requirements for Basic Safety and Essential Performance.
💻 IEC 60601-1-2:2020. Collateral Standard: Electromagnetic Disturbances – Requirements and Tests.
🏡 IEC 60601-1-11:2015. Collateral Standard: Requirements for Medical Electrical Equipment and Medical Electrical Systems Used in the Home Healthcare Environment.
🔧 IEC 60601-1-6:2020. Collateral Standard: Usability.
👩⚕️ IEC 62366-1:2015. Medical Devices – Part 1: Application of Usability Engineering to Medical Devices.
💡 IEC 60601-2-57:2011. Medical Electrical Equipment – Part 2-57: Particular Requirements for the Basic Safety and Essential Performance of Non-Laser Light Source Equipment Intended for Therapeutic, Diagnostic, Monitoring And Cosmetic/Aesthetic Use.
🖥️ IEC 62304:2006. Medical Device Software. Software Life Cycle Processes.
🛡️ IEC 60529:2020. Degrees of Protection Provided by Enclosures (IP Code).
🩺 ISO 13485:2018. Medical Device Manufacturer Certification.
*Test reports available upon demand

Medical and Home Use Certifications
For ISO:13485 Medical Device Manufacturer Certification:
For USA FDA registration details:
For Thai FDA registration details:
For details on EU Declaration of Conformity (DoC) pertaining Electrical CE Mark:
Instructions:

How FlexBeam is different to other devices
- Most Powerful device: power and depth of penetration
- Targeted: by specific placement on the body and by choosing a program associated with the penetration depth
- Wavelength combination: skin surface absorption requires red light and deep penetration requires near-infrared light.
- Flexibility: easily reaches and wraps around most areas of the body, including legs, arms, torso, neck, back, hips, feet, wrists, and more. The device also comes equipped with three sets of easy-to-use, durable double velcro bands to use your FlexBeam anywhere on the body.
- Safety features
- LED-driven, meaning inherently safer than lasers, with no known side-effects
- Auto-shutdown features include:
- Overheat auto shutdown feature (65 C/ 149 F)
- Overcurrent auto shutdown feature (3.5 A)
- 10-minute cycle timer to prevent excessive exposure
- Battery low voltage lockout (9.8V)
How FlexBeam compares to other forms of treatment
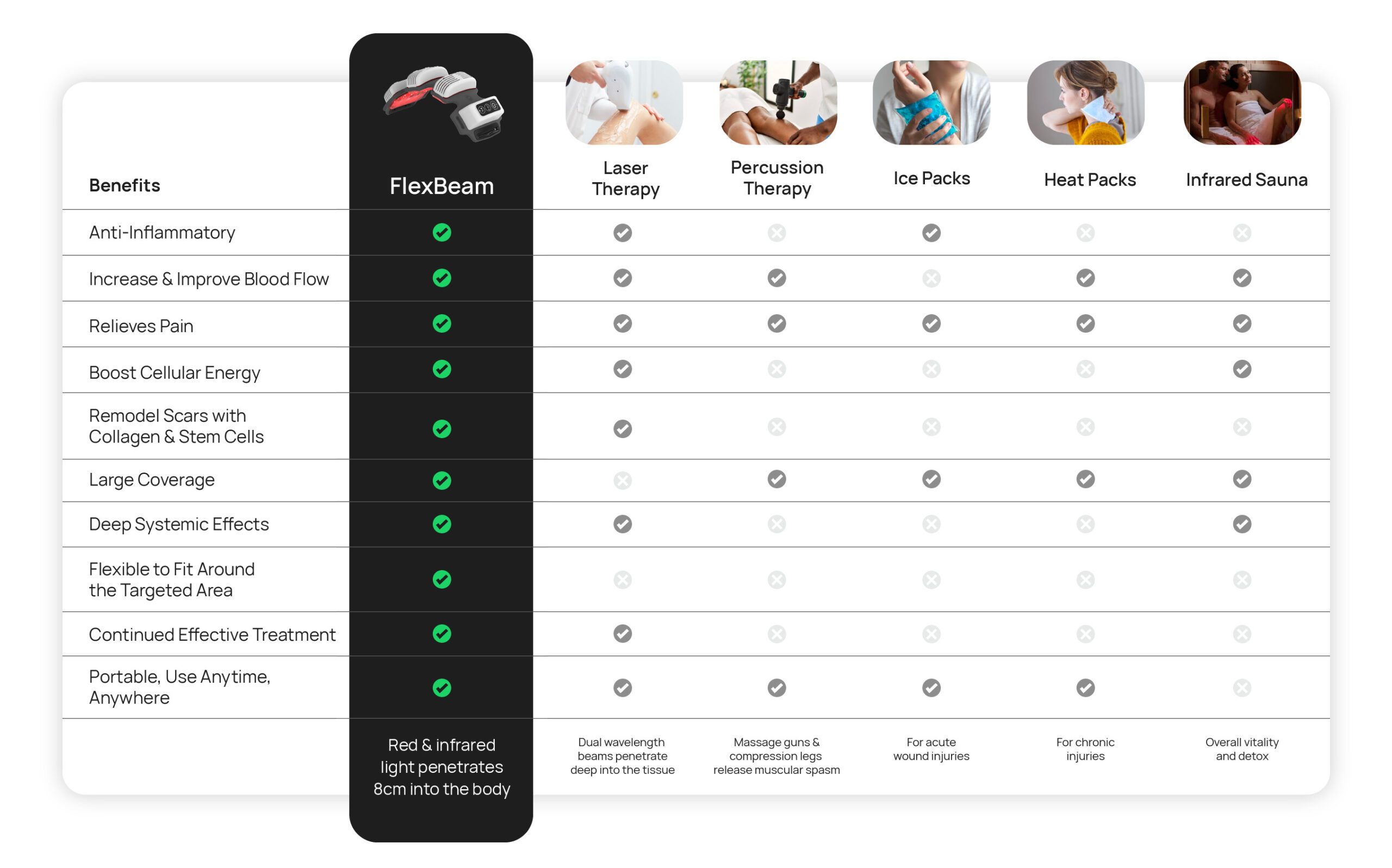

Limited time offer for health & sport professionals
At Recharge Health, we value your reputation as a healthcare and sports practitioner, which is why FlexBeam comes with a 60-day money-back guarantee and a 5-year warranty. Want more units? Write our B2B team by clicking below.
Limited time offer for health & sport professionals
At Recharge Health, we value your reputation as a healthcare and sports practitioner, which is why FlexBeam comes with a 60-day money-back guarantee and a 5-year warranty. Want more units? Write our B2B team by clicking below.
Send us an e-mail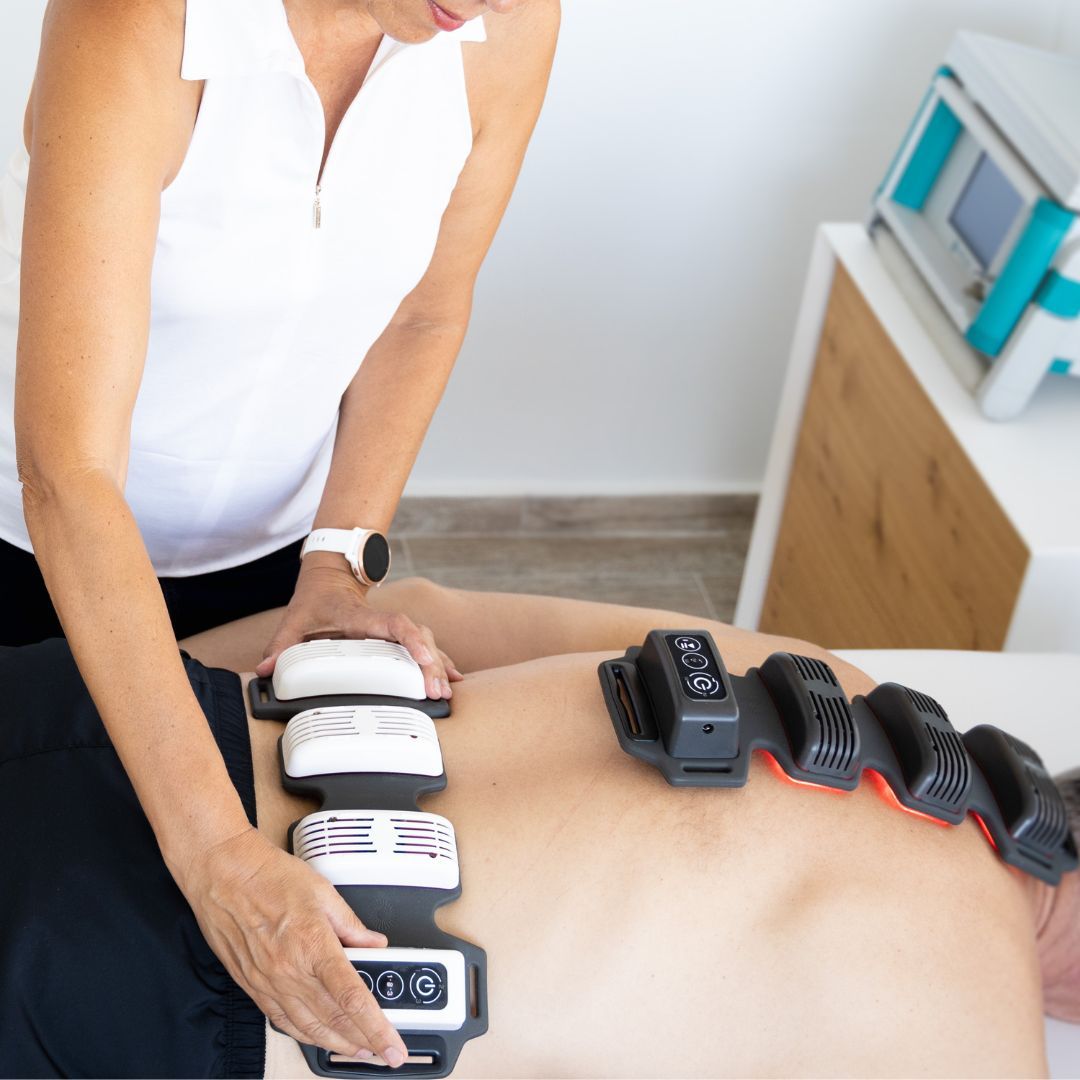
Expand your revenue streams by becoming a reseller or distributor
Want to learn more?
Contact our B2B sales team at [email protected].
Customer Support
Best in class customer support to ensure all of your questions are answered in a timely manner. Issues with a device? Always feel free to reach out. Our team will make sure you’re taken care of.
Safety first
20,000+ devices sold globally without any adverse effects. USA FDA Register and Exempted. FSA/HSA Eligible with TrueMed. IEC and CE Electrical Safety Compliance. Trusted by Top European Athletes and American Physios.
Warranty
5-year warranty for any manufacturing defect. 2-year warranty on battery. Additionally, we’ll replace batteries or devices, when needed. We have two service centers in the UK and EU. Device refurbishment service is available on request.

Get started today
Join the growing community of physical therapists, businesses, and other partnerships sharing the power of red and infrared light with their clients, patients, and customers. Learn more about our tiered approach to wholesale discounts. Email us at: [email protected] or click the button below.
Get started today
Join the growing community of physical therapists, businesses, and other partnerships sharing the power of red and infrared light with their clients, patients, and customers. Learn more about our tiered approach to wholesale discounts. Email us at: [email protected] or click the button below.
Order now